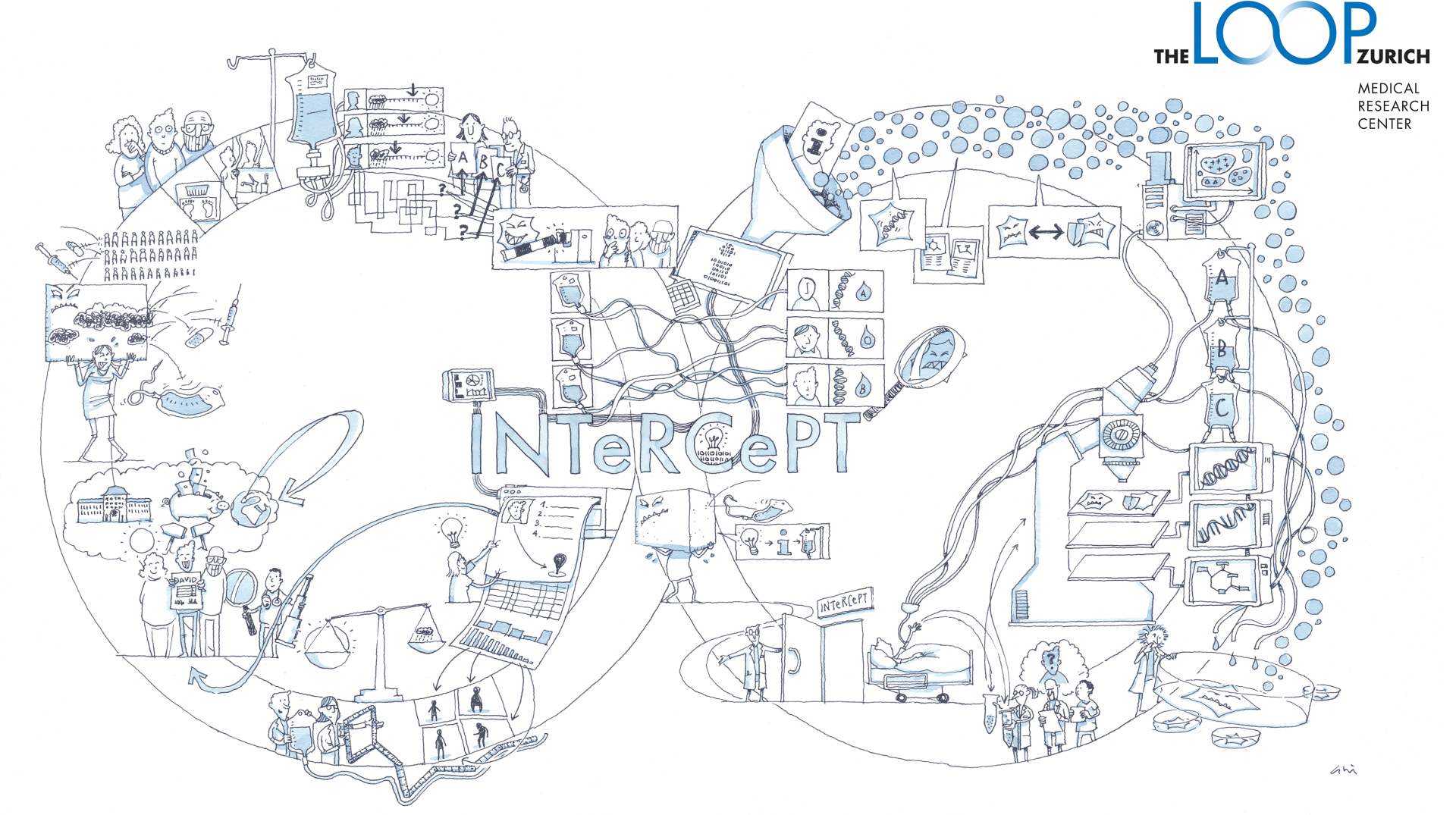
INTeRCePT: Intercept clonal evolution to overcome treatment resistance in childhood and adult blood cancer
Problem: High relapse rate and development of therapy resistance in blood cancer treatment.
Goal: Reduce relapse rate through optimized treatment strategies based on the molecular signature of tumor and immune cells.
The research team led by Thorsten Zenz aims to determine which therapies work best for both children and adults with blood cancer. To this end, an “Innovation Clinic” is being established in Zurich for patients affected by relapse. Analyses of tumor material and healthy blood cells make it possible to create a detailed map of therapy response at the single-cell level. This enhances the understanding of how tumor and healthy immune cells behave and respond to various treatments.
For use in clinical practice, a simplified testing method is being developed – the so-called “INTeRCePT Assay”. This method is designed to provide all the key information that oncologists need for treatment decisions, despite its reduced complexity. The assay is being validated in clinical studies and tested for practical applicability. The goal of this novel precision oncology approach is to increase patient therapy response rates by 50%.
Selected research findings
An ex vivo testing program was developed that allows tumor samples to be studied over time at the single-cell level under the influence of drug treatments. This enables the dynamic observation of both intracellular and extracellular processes as well as their responses to drug therapy. As a result, the role of clonal drivers of tumor growth and the interactions between tumor and immune cells can be better understood.

By analyzing the DNA of individual lymphoma cells, the clonal structure of the tumor cells could be precisely described. These insights help improve the understanding of genetic data and the effects of medications on these cell clones. Based on these findings, and in combination with other so-called multi-omics analyses, drug treatments were individually tailored. Currently, further studies and analyses are underway to continue optimizing these approaches.
The results of the research conducted so far form the basis for the development of the INTeRCePT Assay, a predictive testing method of low complexity, which is to be used in clinical studies. Initial results will be published shortly, while in parallel, ongoing data analyses are being conducted to integrate multi-omics data. The next step is to verify these results in clinical trials.
Clinical studies
In this context, a dataset was compiled containing over 300 samples from 40 lymphoma patients who had received various treatments — including CD19 CAR T-cell therapy. Because samples were collected at multiple time points both before and after the CAR T-cell treatment, a unique dataset was created that allows for the dynamic tracking of changes in immune responses and the tumor microenvironment.
Using advanced biomedical informatics analyses, new therapeutic strategies are now being developed to improve the effectiveness of CAR T-cell therapy. These analyses are expected to be completed within the next year.
Due to recent adjustments in the standard therapy for B-cell non-Hodgkin lymphomas, the study design was revised in consultation with the scientific advisory board. The focus is now on the validation of the INTeRCePT Assay. This represents an important breakthrough that will form the basis for future studies and optimized treatment strategies, laying the groundwork for more targeted interventional studies.
Outlook
In the next twelve months, ongoing experiments and analyses are scheduled to be completed. A key focus will be to gain a deeper understanding of the immune landscape in non-Hodgkin lymphomas. Using multi-omics analyses, the project aims to identify prognostic biomarkers and target molecules for new therapeutic interventions. The INTeRCePT Assay will help increase the efficiency of these analyses and enable better classification of patient groups in future studies, thereby facilitating more targeted therapies.
In parallel, standardized structures for clinical trials and a secure data system are being developed for the Innovation Clinic. Data from more than 300 registered patients – including treatment histories and biobank data – will be documented and made available for research purposes. This will further drive the development of personalized therapies.
Groups involved:
- Thorsten Zenz:Department of Medical Oncology and Hematology, University Hospital Zurich
- Burkhard Becher: Institute of Experimental Immunology, University of Zurich
- Nico Beerenwinkel: Department of Biosystems Science and Engineering, ETH Zurich
- Jean-Pierre Bourquin: Oncology, University Children’s Hospital Zurich
- Stefanie Kreutmair: Department of Medical Oncology and Hematology, University Hospital Zurich
- Wolfgang Huber: Quantitative Biology and Statistics, EMBL Heidelberg
- Andreas Moor: Department of Biosystems Science and Engineering, ETH Zurich
- Berend Snijder: Department of Biology, ETH Zurich

Personalised Treatment of Blood Cancer

↑ Interview with Prof. Zenz (in German)
Project Overview
Lead:
Prof. Dr. med. Thorsten Zenz, Medical Oncology and Hematology Clinic, University Hospital Zurich
Duration: 2021 – 2026
Universities: ETH Zurich, University of Zurich
Hospitals: University Hospital Zurich, University Children’s Hospital Zurich
Researchers: approx. 25
Partners: EMBL Heidelberg
Patients: 100 (more than 5’000 samples)